New Study Reveals Hidden Cancer Weakness Linked to Vitamin B7 and Glutamine
New Study Reveals Hidden Cancer Weakness Linked to Vitamin B7 and Glutamine
A Hidden Metabolic Weakness Inside Cancer Cells
University of Lausanne has recently become the center of attention after a group of researchers uncovered something subtle yet potentially important about how cancer cells survive under stress. The discovery does not revolve around a brand new drug or a dramatic breakthrough in genetic engineering. Instead, it focuses on metabolism, the quiet internal chemistry that keeps cells alive moment by moment.
At first glance, metabolism sounds technical and distant. However, once you look closer, it becomes surprisingly intuitive. Every cell is constantly deciding how to use available nutrients, much like a small city managing electricity, fuel, and raw materials. Cancer cells, though, operate under unusual pressure. They divide rapidly, consume enormous resources, and often survive in environments where nutrients are inconsistent or scarce.
Because of this, scientists have long searched for metabolic weaknesses in tumors. If a cancer cell depends too heavily on one nutrient, removing that nutrient could slow or stop its growth. One of the most studied examples is glutamine, an amino acid that many tumors rely on almost obsessively.
For years, researchers assumed that targeting glutamine metabolism would be a reliable strategy. Yet real clinical outcomes have been mixed. Some tumors respond. Others barely react. The new study helps explain why.
Understanding the Glutamine Dependence of Cancer Cells
Inside every living cell, nutrients are transformed into energy and structural building blocks. Among these nutrients, glutamine plays an unusually versatile role. It contributes nitrogen for protein synthesis and supports the creation of DNA components. It also feeds into energy producing pathways located in the mitochondria.
Cancer cells tend to consume glutamine at accelerated rates. Researchers sometimes describe this behavior informally as glutamine addiction. The term sounds dramatic, but it captures something real. Without glutamine, many tumor cells struggle to continue dividing.
However, biology rarely stays simple for long.
Cells are adaptive systems. When one pathway becomes unavailable, alternative routes often appear. Imagine a busy highway closing during rush hour. Traffic does not stop entirely. Cars reroute through side streets. The flow changes, but movement continues.
Something similar happens in cancer metabolism.
The research team from Molecular Cell explored how certain tumors continue growing even when glutamine is restricted. Instead of collapsing, these cells switch metabolic direction and rely more heavily on carbon based molecules, particularly pyruvate.
This shift may sound minor, yet it reveals a deeper level of metabolic flexibility than previously appreciated.
Pyruvate Steps Into the Spotlight
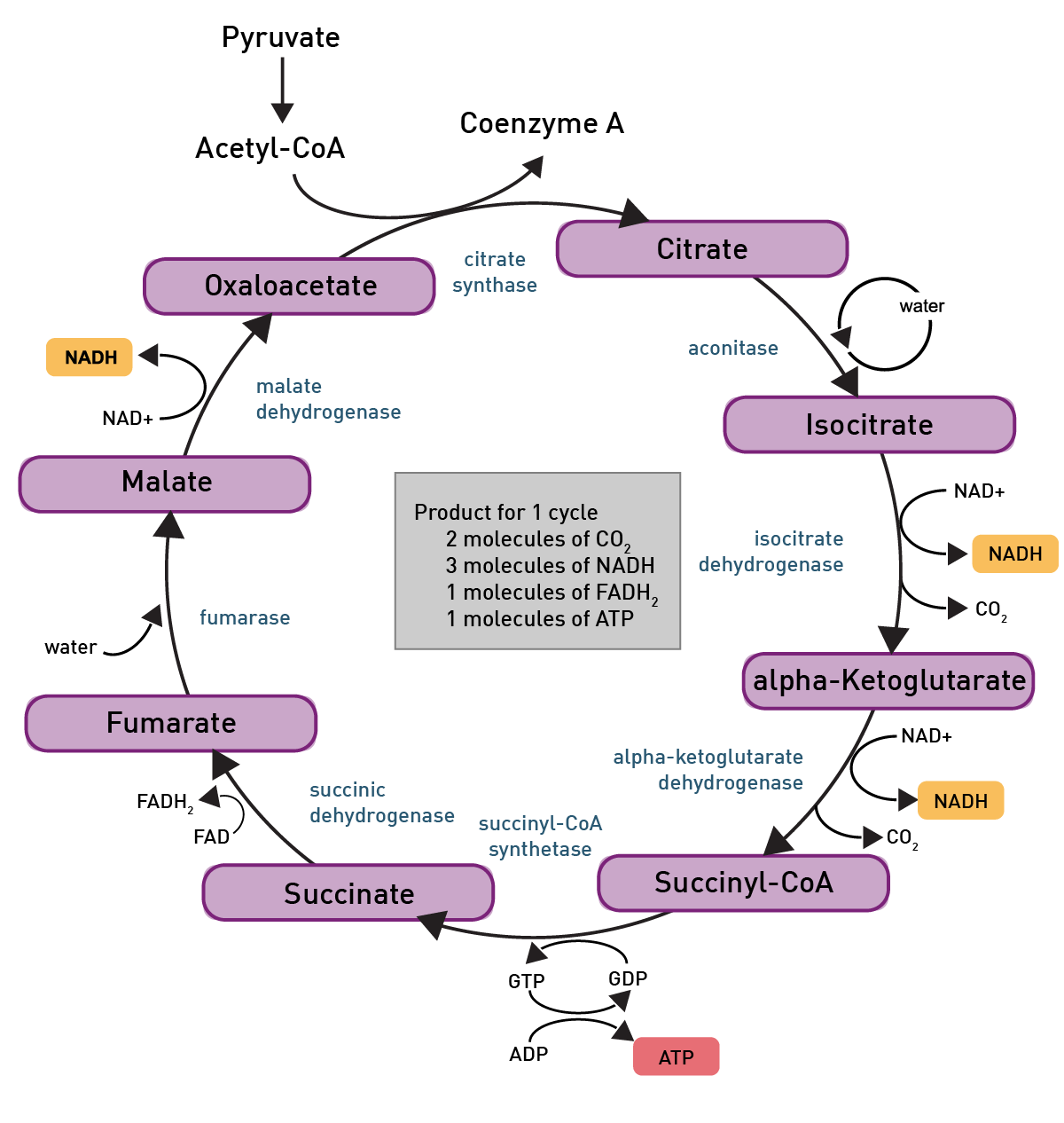
To understand the significance of pyruvate, it helps to picture the broader energy network inside a cell.
Glucose, the sugar commonly discussed in nutrition, is broken down through glycolysis. One of the end products of that process is pyruvate. Normally, pyruvate feeds into the mitochondrial energy cycle or converts into other molecules depending on the cell’s needs.
In tumors lacking glutamine, pyruvate becomes far more important. It essentially compensates for the missing nutrient, allowing the cell to maintain enough metabolic flow to keep dividing.
The research group led by Alexis Jourdain, with major experimental contributions from Miriam Lisci, discovered that this adaptation depends heavily on a specific enzyme called pyruvate carboxylase.
Enzymes function like molecular machines. Without them, chemical reactions slow dramatically or stop altogether. Pyruvate carboxylase allows pyruvate to enter pathways that replenish essential metabolic intermediates. This process becomes especially important when glutamine is absent.
Yet the enzyme cannot function alone.
It requires a helper molecule that might surprise many people.
The Unexpected Role of Vitamin B7
Vitamin B7, also known as biotin, is usually associated with hair supplements or general nutrition advice. It rarely appears in conversations about cancer biology. Nevertheless, in this metabolic context, biotin plays a critical role.
Pyruvate carboxylase requires biotin in order to function properly. Without it, the enzyme remains inactive, almost like an engine without ignition.
The researchers observed that when cells lacked vitamin B7, the pyruvate pathway collapsed. Even if pyruvate itself was present, the metabolic compensation failed. Cell division slowed dramatically.
This discovery introduces an interesting layer of nuance.
Cancer metabolism is often described using broad categories such as sugar metabolism or amino acid metabolism. However, the reality is far more granular. Tiny molecular dependencies, including vitamins, can shift the entire balance of growth and survival.
At the same time, it would be misleading to interpret this finding too quickly as a simple dietary solution. Reducing vitamin intake does not automatically translate into effective cancer treatment. Human physiology is interconnected, and normal cells also require these nutrients.
Still, the observation opens new conceptual doors.
Instead of targeting a single nutrient like glutamine, researchers might need to consider entire metabolic networks.
The Genetic Layer That Changes Everything
Metabolism alone does not tell the whole story. Genes shape how metabolic pathways operate, and mutations can dramatically alter cellular behavior.
One gene examined in the study is FBXW7, which has already been linked to multiple cancer types. Mutations in this gene appear frequently in tumor samples, although their precise metabolic consequences were not fully understood.
The research team discovered something intriguing.
When FBXW7 is mutated, levels of pyruvate carboxylase decrease. As a result, the cell loses its ability to effectively use pyruvate as a backup fuel source. Without this alternative route, the tumor becomes more dependent on glutamine again.
This creates a kind of metabolic bottleneck.
Cells with certain FBXW7 mutations cannot easily switch pathways. Their flexibility narrows. From a therapeutic perspective, that limitation could become a vulnerability.
The experiments linking these mutations to metabolic behavior involved collaboration with additional research groups, including scientists at Northeastern University and proteomics specialist Owen Skinner.
By combining metabolomics and proteomics data, the team could observe both chemical changes and protein level shifts. This multi layer approach helps avoid oversimplified interpretations.
Why Some Anti Glutamine Treatments Fail
For years, drug developers have attempted to block glutamine metabolism directly. The logic seemed straightforward. If cancer cells need glutamine, removing it should stop tumor growth.
Reality proved more complicated.
Some tumors adapted. Others did not rely on glutamine as heavily as expected. Clinical responses varied widely.
The new findings help explain this inconsistency.
When pyruvate pathways remain functional, cancer cells can compensate for reduced glutamine. They reroute metabolic flow and continue dividing. However, when genetic mutations or vitamin related constraints disrupt that backup system, glutamine dependence becomes stronger.
This variability may explain why certain patients respond to metabolic therapies while others see limited benefit.
In practical terms, it suggests that treatment strategies may need personalization based on metabolic profiling rather than relying solely on tumor type.
The concept is not entirely new, yet this study adds concrete biochemical detail.
Metabolic Flexibility as a Survival Strategy
One of the most fascinating aspects of cancer biology is adaptability.
Tumors evolve under pressure. Chemotherapy, immune responses, and nutrient limitations all push cancer cells to adjust their internal processes. Over time, resistant populations emerge.
Metabolic flexibility plays a major role in that process.
Instead of using a single energy source, cancer cells often maintain several options simultaneously. When one pathway weakens, another strengthens. This layered redundancy makes treatment challenging.
Consider an everyday analogy.
If a household depends entirely on electricity and the power fails, everything stops. However, if that same household also has solar panels and a backup generator, daily activity continues with minimal disruption.
Cancer cells operate in a similar way, though at a microscopic scale.
The discovery involving pyruvate and vitamin B7 reinforces this idea. Blocking one nutrient is rarely enough because alternative routes exist.
Therefore, future therapies may need to target multiple metabolic pathways at once.
A More Realistic View of Nutrient Based Therapies
There is often public excitement when research mentions vitamins or dietary molecules in connection with cancer. Understandably, people hope that simple nutritional changes might offer protection or treatment.
However, the reality is more complex.
Inside the human body, nutrients circulate through many tissues simultaneously. Any attempt to manipulate vitamin levels must consider effects on healthy cells as well.
Biotin, for example, is essential for normal metabolism. Severe deficiency would create systemic problems long before affecting tumor growth in a controlled way.
Therefore, researchers are not suggesting that vitamin restriction alone could treat cancer. Instead, they are identifying biochemical dependencies that may guide drug design.
A future therapy might target the enzyme system directly rather than altering dietary intake.
This distinction matters because it prevents oversimplified conclusions.
The Importance of Integrated Research Approaches
Another valuable aspect of this study lies in its methodology.
Modern cancer research increasingly combines several analytical layers. Genomics identifies mutations. Proteomics tracks protein abundance. Metabolomics measures chemical flow inside cells.
Individually, each approach provides partial insight. Together, they reveal a more complete picture.
In this case, genetic data linked FBXW7 mutations to enzyme regulation, while metabolomic analysis demonstrated how nutrient pathways shifted under stress.
This integrated approach reflects a broader trend in biomedical science. Complex diseases rarely yield to single method explanations.
Moreover, interdisciplinary collaboration is becoming standard rather than exceptional.
Where the Research May Lead Next
Although the findings are compelling, they represent an early stage of understanding rather than an immediate clinical solution.
Several questions remain open.
For example, do all tumor types rely equally on the pyruvate pathway when glutamine becomes scarce. Are certain cancer environments more dependent on biotin mediated enzyme activity. How do other genetic mutations interact with these metabolic routes.
Future studies will likely explore these areas.
Researchers may also attempt to develop compounds that selectively disrupt pyruvate carboxylase activity in tumor cells without harming normal tissues. Achieving that balance remains one of the central challenges in oncology.
There is also growing interest in adaptive treatment models, where therapy changes dynamically based on tumor metabolism over time.
Cancer is not static. Treatments that account for metabolic evolution may prove more effective.
A Subtle but Meaningful Step Forward
Scientific progress often arrives quietly.
Instead of dramatic breakthroughs that immediately transform medicine, many advances refine understanding piece by piece. This study fits that pattern.
It does not claim to cure cancer. It does not promise rapid therapeutic application. Yet it clarifies an important detail about how tumor cells survive metabolic stress.
That clarity matters.
When researchers understand why certain treatments fail, they gain direction for improving them. In this case, the discovery highlights the interplay between nutrients, enzymes, and genetic mutations.
It also reinforces a broader lesson.
Cancer is not defined by a single weakness. It is defined by a network of conditional vulnerabilities that shift depending on context.
Thinking About Cancer Metabolism in a More Human Way
There is something almost philosophical about metabolic research.
At a microscopic level, cells are constantly negotiating survival. They adjust resources, adapt to constraints, and respond to environmental uncertainty. In many ways, the behavior mirrors larger systems we see in everyday life.
When one resource disappears, alternatives emerge. When flexibility narrows, vulnerability increases.
The study from the University of Lausanne captures that dynamic clearly.
Pyruvate becomes a backup plan. Vitamin B7 becomes a silent enabler. A gene mutation shifts the entire balance.
None of these elements alone tells the full story. Together, they reveal how intricate biological survival strategies can be.
And perhaps that is the most interesting takeaway.
Cancer research is not only about destroying cells. It is about understanding how life persists under pressure, even when that persistence becomes harmful.
The more precisely scientists map these survival pathways, the closer medicine moves toward treatments that are both smarter and more targeted.
Progress may be gradual, but each layer of understanding builds momentum.
Open Your Mind !!!
Source : SciTechDaily








Comments
Post a Comment